How is IIT Bombay Revolutionizing T-cell Recovery for Cancer Treatment?
Synopsis
Key Takeaways
New Delhi, Feb 4 (NationPress) Scientists at the Indian Institute of Technology (IIT) Bombay have pioneered a more streamlined and effective approach to recover immune cells cultivated in the laboratory for T-cell–based cancer treatments.
In therapies like CAR T-cell, T-cells (a specific kind of immune cell) are extracted from a patient’s blood, modified extensively in the lab, and then reintroduced into the patient's bloodstream to combat cancer.
These cells, bred externally, must be harvested delicately to ensure they remain viable and functional upon reintroduction to the patient. Thus, identifying safe, effective methods for T-cell cultivation and retrieval is crucial for the success of these therapies.
“While cell recovery may seem straightforward in theory, it presents significant challenges in reality,” remarked Prof. Prakriti Tayalia from the Department of Biosciences and Bioengineering at IIT Bombay.
“Insufficient healthy cells hinder both proper testing and therapeutic application,” she added.
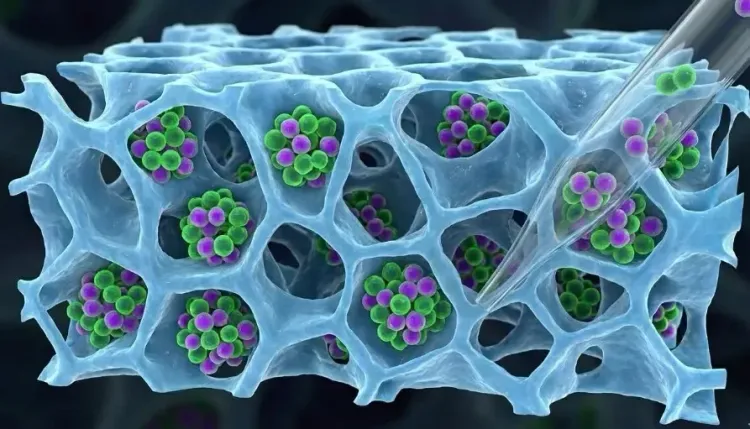
To better replicate the body’s natural environment, Tayalia’s team utilized a specific type of scaffold created through a process known as electrospinning. These electrospun scaffolds resemble thin mats of extremely fine fibers, akin to a dense fishing net.
The team cultivated Jurkat T-cells (a human cell line utilized in laboratory studies for T-cell biology, cancer, and HIV) within electrospun scaffolds made from a material named polycaprolactone.
Under microscopic observation, the researchers noted that the cells actively migrated into the scaffold and became securely lodged between the fibers.
Furthermore, the research revealed that using trypsin, an enzyme, for cell collection resulted in higher cell mortality.
Conversely, cells retrieved with accutase, a gentler enzyme, exhibited greater survival rates and functioned more like healthy T-cells. They formed clusters, a crucial precursor to T-cell division, and continued to thrive post-recovery.
“Aggressive treatments involving enzymes like trypsin can damage vital surface proteins needed for immune signaling and activation, thereby diminishing the therapeutic potential of the cells. Accutase appears gentle enough to prevent this issue,” Tayalia explained.
The findings of this study, published in the journal Biomaterials Science, could assist laboratories in utilizing such scaffolds when preparing cells for therapies like CAR T-cell treatment.
“Every step is critical if we want these advanced therapies to benefit patients. The methods used to grow and retrieve cells can significantly impact outcomes,” Tayalia emphasized.