India Initiates Groundbreaking Clinical Trial for Ayurveda in TB Treatment
Synopsis
Key Takeaways
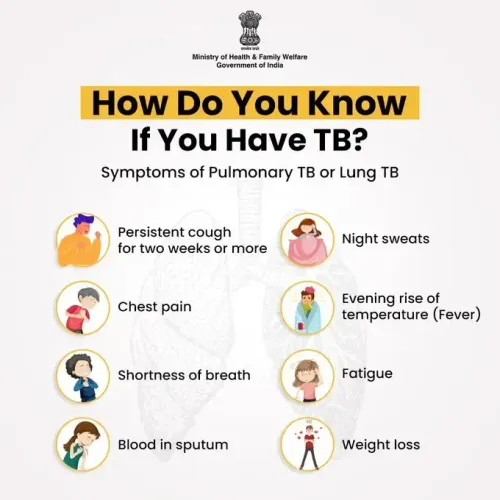
New Delhi, March 24 (NationPress) In a groundbreaking initiative on World Tuberculosis Day, India has launched the first-ever clinical trial globally to rigorously assess the role of Ayurveda as a supplementary treatment alongside standard Anti-Tuberculosis Treatment (ATT), as announced by the government on Tuesday.
This pioneering clinical trial, a collaborative effort between the Department of Biotechnology and the Ministry of Ayush, aims to enroll 1,250 newly diagnosed tuberculosis patients across eight institutions. The study will investigate how Ayurveda can complement standard treatment, focusing on key factors such as body weight, nutritional outcomes, disease progression, quality of life, safety, and tolerability.
Union Minister of State for Science & Technology, Jitendra Singh, emphasized that this study embodies the spirit of a “whole-of-science” approach by integrating the fields of biotechnology and Ayurveda, and reflects a “whole-of-government” strategy through inter-ministerial collaboration.
According to Prof. Vaidya Rabinarayan Acharya, Director General of CCRAS, the initiative has successfully moved through various phases including consultations, protocol development, and necessary approvals.
Dr. Debasisa Mohanty, Director of BRIC-NII, explained that the study will explore tuberculosis-associated cachexia as an immune-metabolic condition, utilizing cutting-edge techniques such as DEXA, MRI, immune profiling, metabolomics, and single-cell RNA sequencing to assess changes in body composition, immune function, and energy metabolism.
The objective is to determine how integrative interventions can bolster recovery and influence long-term outcomes, as stated by the Ministry of Science & Technology.
Minister Singh noted that India, which bears nearly 25% of the global TB burden, has achieved a 21% reduction in tuberculosis incidence over the past decade, with current rates at about 187 cases per 100,000 population as of 2024.
“This decline in TB incidence is a testament to India’s committed and innovative efforts. Together, we will strive for a TB-free India,” he quoted Prime Minister Narendra Modi.
India has set forth an ambitious and expedited agenda towards TB elimination, enhancing early diagnosis, universal drug susceptibility testing, digital adherence technologies, and patient-centric care under the National TB Elimination Programme, the minister highlighted.
He also pointed out the interconnectedness between TB and other health conditions like diabetes, underscoring the necessity of integrated strategies for effective disease management.
Furthermore, he mentioned the RePORT India initiative, one of the largest TB research consortia, which includes over 4,500 enrolled TB patients and more than 5,000 household contacts, producing evidence crucial for global policy frameworks, including WHO guidelines on nutrition and tuberculosis.
aar/pk