Is a New Monoclonal Antibody Treatment Safe and Effective for Rare Liver Disease?
Synopsis
Key Takeaways
New Delhi, Dec 24 (NationPress) A novel monoclonal antibody therapy has demonstrated encouraging outcomes for a rare liver condition known as primary sclerosing cholangitis (PSC). The research team from the University of California-Davis, US, evaluated an anti-inflammatory and anti-fibrotic monoclonal antibody termed nebokitug, confirming its safety and potential efficacy in PSC patients.
As reported in the American Journal of Gastroenterology, these findings bring hope to individuals suffering from PSC, which currently lacks any effective treatments aside from liver transplantation.
"During the trial, nebokitug revealed its potential to transform the lives of PSC patients by alleviating fibrosis and inflammation, ultimately leading to better health outcomes," stated Christopher Bowlus, the head of Gastroenterology and Hepatology at UC Davis Health.
"This is excellent news for PSC patients who urgently require an efficient, FDA-approved treatment option."
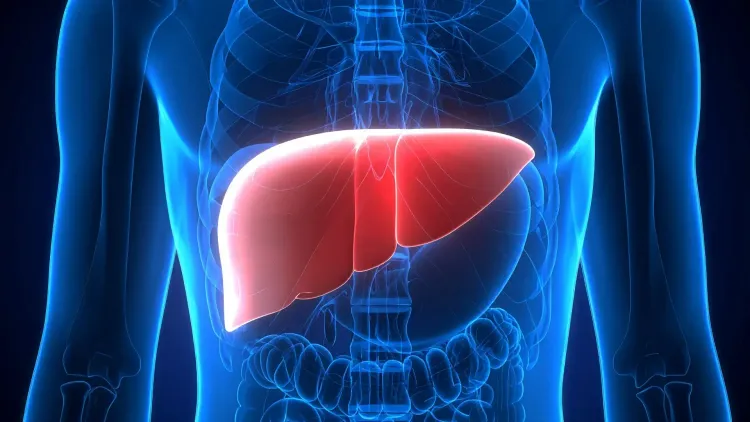
PSC is a rare, chronic liver disease characterized by inflammation and damage to the bile ducts, which transport bile from the liver to the small intestine to assist in fat digestion. When these ducts suffer injury and narrow, bile accumulates in the liver, resulting in long-term liver damage.
While the exact etiology of PSC remains unclear, many patients also experience inflammatory bowel disease, indicating a potential correlation between gut inflammation and liver health.
Common symptoms include fatigue, itching, and jaundice, though some individuals may not exhibit any symptoms initially. There is no known cure, and treatments primarily aim to alleviate symptoms and related complications. In severe cases, a liver transplant may be necessary.
Nebokitug is a laboratory-engineered antibody designed to inhibit a protein called CCL24, which contributes to inflammation and scarring through its interaction with specific inflammatory cells in the liver.
In patients with PSC, CCL24 levels are elevated and localized around the bile ducts, exacerbating liver damage. Research has indicated that blocking CCL24 can mitigate these detrimental effects.
In the Phase 2 clinical trial, 76 PSC patients from five different countries were randomly assigned to receive nebokitug at two varying doses or a placebo intravenously every three weeks for a duration of 15 weeks, primarily to assess safety.
The findings indicated that nebokitug was safe and well-tolerated. Notably, patients suffering from advanced liver scarring displayed improvements in key indicators such as liver stiffness and fibrosis markers compared to those receiving the placebo.