Could the AIIMS-led Trial of the Most Advanced Brain Stent Revolutionize Stroke Treatment?

Synopsis
Key Takeaways
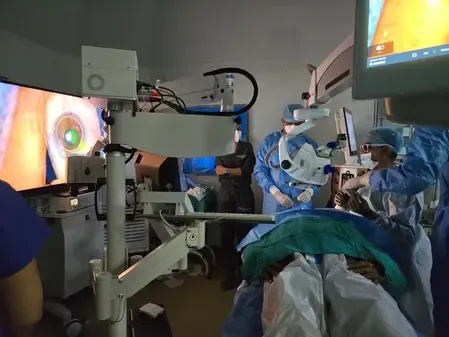
New Delhi, Dec 13 (NationPress) The Supernova Stent — an innovative brain treatment device — has been proven to be both safe and effective for stroke patients, as reported by specialists at AIIMS Delhi, who spearheaded the inaugural clinical trial.
AIIMS Delhi served as the national coordinating center and the primary enrolling site for the GRASSROOT trial involving the Supernova Stent.
“This trial marks a pivotal moment for stroke treatment in India,” stated Dr. Shailesh B. Gaikwad, Professor and Head of the Department of Neuroimaging & Interventional Neuroradiology at AIIMS Delhi, and National Principal Investigator of the GRASSROOT Trial.
“According to preliminary trial results, the Supernova stent has exhibited outstanding safety and efficacy outcomes in the management of severe strokes,” he elaborated, referencing findings published in the esteemed Journal of Neurointerventional Surgery (JNIS).
In the initial prospective multicenter thrombectomy trial, the Supernova stent retriever achieved a high rate of successful blood flow restoration, with a brain bleed rate of just 3.1 percent, a mortality rate of 9.4 percent, and 50 percent functional independence at 90 days.
Developed by Gravity Medical Technology, the Supernova is tailored for India's diverse patient demographics, where strokes frequently affect younger individuals compared to Western countries.
Earlier this year, data from the GRASSROOT trial received approval from the Central Drugs Standard Control Organisation (CDSCO), allowing the Supernova stent-retriever to be utilized routinely in India.
The GRASSROOT India trial, which verified the device's safety and efficacy in addressing life-threatening strokes, was conducted across eight centers. Experts note that this trial represents a significant achievement for the Make-in-India initiative, establishing India as a key player in advanced stroke care.
“The device has already treated over 300 patients in Southeast Asia and will soon be manufactured domestically, making it available in India at affordable prices, thereby providing new hope to the 1.7 million Indians affected by strokes annually,” said Dr. Dileep Yavagal, Professor of Neurology and Neurosurgery at the University of Miami, involved in the global trial.